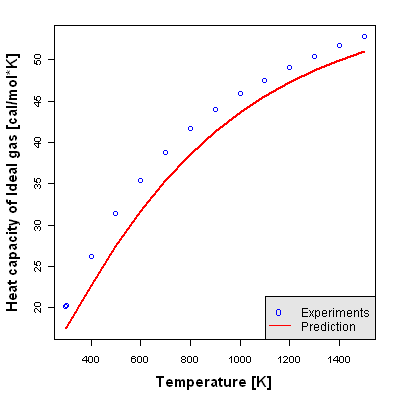
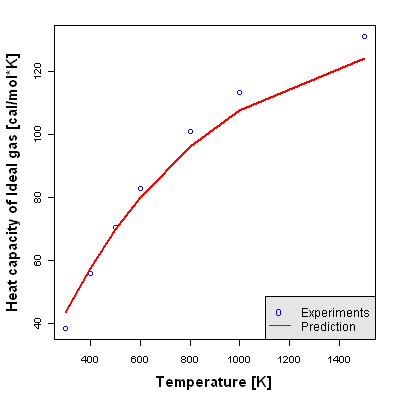
Heat Capacity of Ideal Gas
Definition
Heat Capacity of Ideal Gas is defined as the calorie required to increase the temperature of 1mol by 1K, assuming that a pure substance is an ideal gas.
Estimation Method
MolInstincts
Property values are estimated by the combination of fundamental thermodynamic theories, modern modeling approaches, e.g., SVRC (Scaled Variable Reduced Coordinates), QSPR (quantitative structure-property relationships) methods, and ANN (Artificial Neural Network) with over fitting prevention algorithm, utilizing the quantum chemical calculations results and molecular descriptors. The estimated value is finally determined after manual inspection.
Joback
Group-contribution method (Estimation of pure-component properties from group-contributions, Chem.Eng.Commun., 1987. 57. 233)
※ The values by the previously known prediction method is obtained by using constant property values forecasted from MolInstincts as calculation factors.
Examples for each Comment
Accuracy Level 1
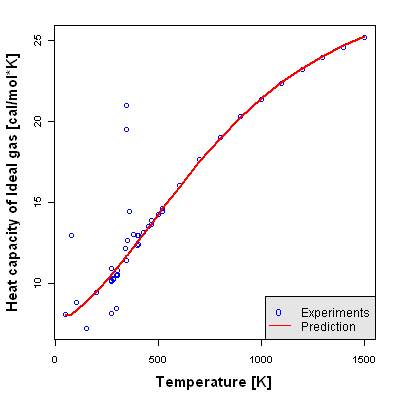
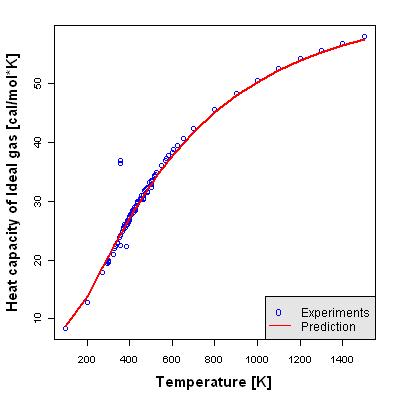
Accuracy Level 2
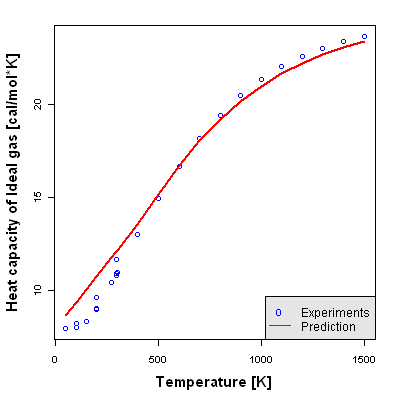
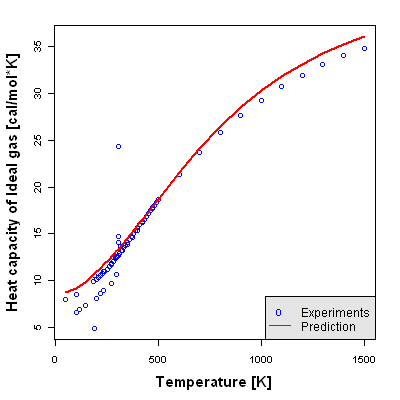
Accuracy Level 3